- Products
- Service & Help
 Our printing experts will be happy to help you with any questions you may have about your order. Simply use our customer support.SupportNewsletter
Our printing experts will be happy to help you with any questions you may have about your order. Simply use our customer support.SupportNewsletter - About us
 Our printing experts will be happy to help you with any questions you may have about your order. Simply use our customer support.SupportNewsletter
Our printing experts will be happy to help you with any questions you may have about your order. Simply use our customer support.SupportNewsletter - Contact us
- Premium service
- Sustainability
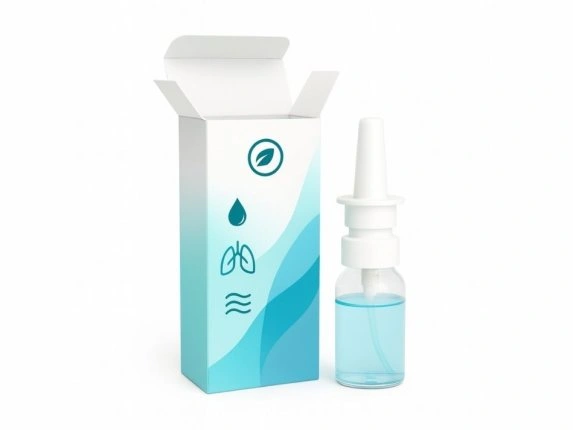
Pharmaceutical spray & inhaler packaging
Protect and enhance the presentation of your nasal sprays, oral sprays, or inhaler devices with our custom-designed cardboard packaging, tailored to the pharmaceutical industry's requirements. Our boxes offer a reliable solution for the secondary packaging of your medical devices, whether metered-dose sprays, pressurised bottles, or mouthpiece inhalers. Customisable, robust, and recyclable, they guarantee a professional presentation that complies with European standards. Labelprint24 supplies exclusively the cardboard packaging – not the devices themselves – with fast, 100% European manufacturing and comprehensive support.
Discover our complete range of packaging at Labelprint24.com!
- ProductionFrom 3 working days
- MaterialsGC1 / GC2 / GD2
- SizeCustom size
- OptionsLaminate / Varnish / Clear window
- Online DesignerCustom Design
- Printing TechnologyDigital / Offset



Ronan Joncour
Product consultant Folding Boxes
Whether it’s a simple box or a folding carton with an inlay – I’m your advisor for carton types, base designs, and finishing options. From cosmetics to food and technology, I’ll help you find the perfect folding carton for your industry.
"Just a carton? Folding boxes are incredibly versatile!"
I will be happy to help you personally.
Why choose our spray packaging & inhaler packaging?
Our packaging is specially designed for the complex formats of sprays and inhalers. Each box is fitted precisely to ensure the device's stability during transport and offers intuitive and secure opening for the end user.
Medical professionals value our packaging for its technical cutouts for nozzles, safety clips, or multilingual instructions, transforming it into a vital communication tool in pharmacies or direct sales. Additionally, all boxes are printed with low-impact inks on recyclable materials, aligning with CSR requirements.

Materials and finishes available
Choosing the right material is important for the strength and compliance of your spray packaging. At Labelprint24, we use folding carton, coated carton, or recycled carton, with weights from 250 to 500 g/m². This allows us to adapt to the weight of the device and its usage conditions, while ensuring perfect stability on shelves or during transport. Coated carton offers a smooth finish ideal for high-quality printing, while recycled carton provides an aesthetically pleasing and responsible alternative, supporting CSR initiatives.
In terms of finishes, we offer a complete range: matte or glossy lamination to protect and enhance the surface, spot varnish to highlight specific graphic areas, embossing, or foil stamping to enhance your product's the high-end perception. It is also possible to integrate a cut-out window or a specific compartment for a nozzle, safety cap, or folding instruction leaflet. Each finish is designed to combine functionality with visual impact.
Customisation options
Each spray or inhaler has its own specific requirements regarding shape, volume, and handling. That's why we design each cardboard package to be completely customised, taking into account the specific characteristics of your medical device.
You can precisely define the dimensions of your pharmaceutical aerosol packaging based on the height, diameter, and shape of the bottle or inhaler. Our design office then adapts the cardboard cutout to guarantee a perfect fit and optimal protection, even during delicate transport or packaging in pharmacies.
We also offer integrated technical cutouts: a front opening for the nasal applicator, a notch for the dosing cap, an internal compartment for a multilingual leaflet, or cardboard inserts to stabilise the device. These options allow you to create functional packaging designed to facilitate the user experience and help ensure safety.
For printing, you can customise the entire exterior surface of your nasal spray packaging, and even the interior, with your branding, regulatory information, batch numbers, and Datamatrix codes. Our offset or digital printing presses offer high-definition output, compliant with regulations.
Finally, our team will support you at every stage to ensure your design meets regulatory requirements (readability, contrast, technical areas, information hierarchy) and guarantees a 100% ready-to-use product.
Technical specifications
Our MDI inhaler packaging and oral spray packaging is made from industrial-grade materials ensuring durability and compliance with medical industry requirements. The cardboard used, whether folding, coated, or recycled, has a weight between 250 and 500 g/m², ensuring a rigid structure capable of effectively protecting even the most fragile devices.
Each package is custom-designed to the exact dimensions of your device. We produce cases adapted to all types of sprays or inhalers: nasal sprays, sublingual devices, bottles with metered-dose pumps, or pressurised inhalers. We also offer secure closure with either simple or reinforced tabs, with or without a tamper-evident notch.
Printing is available in four-colour offset or digital formats, with the option of printing the inside of the box as well. Available finishes include lamination, spot varnish, hot foil stamping, embossing and custom cuts.
The minimum order quantity starts at 100 units, with production capacities suitable for both small runs and large volumes. Once the proof is approved, the average manufacturing time is 7 to 10 business days. All production is carried out in-house in Europe, with quality control at every stage.
Applications & use cases
Cardboard packaging for sprays and inhalers is suitable for a wide range of applications in the pharmaceutical, parapharmaceutical, medical, and wellness sectors. Our boxes are designed to meet the technical and regulatory requirements related to distribution, patient safety, and brand image.
They are commonly used for the secondary packaging of nasal sprays for the treatment of allergies or rhinitis, as well as for oral or sublingual devices, particularly in the areas of dietary supplements and oral hygiene products. Thanks to their custom design, these packages effectively protect your product while ensuring a professional presentation at the point of sale.
We also work with manufacturers of pressurised metered-dose inhalers (MDIs) for asthma or dry inhalers with mouthpieces used in pulmonology. Our boxes allow for the inclusion of all the necessary components for product use: instructions for use, cap, replacement mouthpiece, etc., while complying with regulatory requirements.
Furthermore, our oral spray packaging and nasal spray packaging is perfectly suited to products intended for e-commerce or direct sales. Their robustness ensures the security of the contents during shipping, while their graphic customisation allows for maintaining a high level of branding, even outside traditional distribution channels.
How to order online
Ordering from Labelprint24 is straightforward via our online platform. In just a few steps, you get pharmaceutical aerosol packaging perfectly suited to your product and ready for production.
Start by defining the exact dimensions of your case based on the size of the bottle or device. Our configurator allows you to precisely adjust the height, width, and depth, and add options such as technical cutouts, windows, or cushioning inserts if needed.
Next, you choose the type of cardboard, the desired printing, the finishes, and the quantities. Once these elements are confirmed, you can upload your graphic file directly to the platform or use our pre-configured templates to create your design.
Each order undergoes prepress checks: our team verifies technical compliance and provides you with a digital proof for approval before production begins. Payment is secure, order tracking is available online, and delivery is guaranteed quickly throughout France and Europe.
FAQs
No. We exclusively produce the secondary cardboard packaging. Sprays, bottles, mouthpieces, or inhaler devices must be supplied by the customer. Our cases are designed to fit these items perfectly.
Yes. Each package is designed entirely to measure, based on the exact dimensions of your product. You can specify these measurements during the online configuration or send them to our technical team.
Absolutely. Our packaging meets European requirements for labeling, traceability, legibility of information, and safety for medical devices. We assist you in validating the compliance of your files.
The minimum order starts at 100 units, with tiered pricing based on volume. We also offer solutions tailored to large industrial production runs.
Yes. Our team can help you adapt your files, create a template, or integrate the regulatory elements necessary to ensure your packaging meets all requirements in. your industry.
After approval of the proof of your MDI inhaler packaging or oral spray packaging, production typically takes 7 to 10 business days. Delivery is fast, with tracking available throughout the UK and Europe.
Your B2B supplier for packaging - all from one provider